Myostatin, or growth and differentiation factor 8 (GDF-8), also called the ‘Hercules gene’, is a protein based hormone, that acts at physiological levels to limit muscle mass. Research over the years in multiple animal species has shown that, inhibition of Myostatin gene promote dramatic increase in muscle mass as a result of the combination of hypertrophy and hyperplasia.
After animals are born, the myostatin gene negatively regulates the growth and development of skeletal muscle by limiting the number and size of muscle fibres.
Myostatin inhibition by genetic manipulation and/or drugs can be a promising strategy to counteract muscle wasting in a diversity of settings.
Myostatin is the most common type of skeletal muscle protein, it is of interest in studies related to sports science. However, its expression is also noted in the blood, heart, lungs, reproductive system and adipose tissue. Emerging evidence suggests that myostatin not only regulates muscle mass, but also metabolism, adiposity and insulin-sensitivity.
Mice studies have shown that, myostatin provides a novel therapeutic option to decrease adipocyte size, prevent obesity and improve glucose tolerance.
Myostatin is produced by the muscle tissue of the heart, and damage to the heart causes it to be released into the bloodstream. Once in the bloodstream, myostatin reaches the muscles and causes them to weaken over time. As a result, patients with heart disease often also experience symptoms of muscle atrophy.
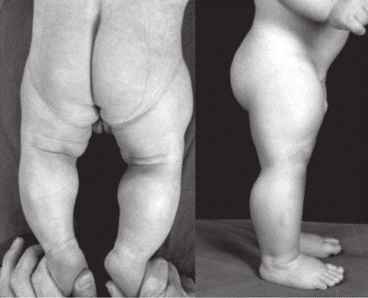
Myostatin deficient animals display an increase in skeletal muscle mass known as double muscling (DBM). For nearly two hundred years, the phenomenon of the “double-muscled” animal, a spectacular model of muscular hypertrophy, has represented a never-ending source of inspiration and argument for both livestock producers and scientists.

The stunning muscularity of such animals embodies the extreme in meat-producing efficiency, each animal yielding a large proportion of retail cuts that are low in fat and characteristically tender. The meat of double-muscled animals is generally much lower in fat, and what fat does remain is higher in the polyunsaturated varieties, both of which more closely conform to current nutritional guidelines.
In spite of the name, a double-muscled animal has the same number of muscles as a conventional animal. Rather, hypertrophy of the muscular tissue and the extreme scarcity of fat cover sharply define every muscle in the animal’s body. For e.g. double-muscled cattle develop more muscle fibres than normal cattle. This cellular hyperplasia is pronounced in the foetal stage of growth in double-muscled cattle, with cell number increasing at a rate nearly three times that of normal cattle.
However, these animals are more susceptible to respiratory disease, urolithiasis, lameness, nutritional stress, heat and dystocia, resulting in lower robustness. Double-muscled cattle have shown signs of fatiguing faster than normal cattle during forced exercise, simply because of increased proportion of fast twitch fibres.
Though, myostatin-related muscle hypertrophy is not known to cause any medical problems, and affected individuals are intellectually normal.
It was only in the late 90s, when the knowledge about myostatin came into picture. This discovery was considered a significant success in the study of genetic factors for increasing muscle mass and developing strength abilities.
In 1997, a research team at Johns Hopkins School of Medicine described Myostatin for the first time as a negative regulator of muscle mass in mammals. With the purpose of determining Myostatin function, researchers disrupted the gene in mice, which then presented significant increase in muscle mass (hypertrophy and hyperplasia). Eventually similar changes were seen in cattle, dogs, pigs, rabbits, poultry & horses.
The first case of a human mutation in the Myostatin gene was described in 2004 study, when a German new-born presented extraordinary musculature (mainly in the arms and legs) compared to a child of the same age. By 4.5 years of age, the child was able to hold two/three kg dumbbells with his arms extended, and muscle hypertrophy was analysed by ultrasound at six years of age, when the cross sectional area was also 2.5 times greater than that of a normal child.
FACTORS AFFECTING MYOSTATIN
Factors affecting Myostatin:
- There are a number of factors that act as inhibitors of myostatin synthesis, including the myocyte 2 enhancing factor (MEF2); hormones, such as insulin-like growth factor (IGF-1), angiotensin II, thyroid hormone, erythropoietin , sex steroids, follistatin, and estradiol.
- One of the main factors in sports that significantly affects the level of myostatin secreted is power-oriented physical activity, hypoxia, and dietary supplements. Moreover, the production of myostatin is influenced by essential amino acids, which are often consumed by athletes.
- Age affects myostatin concentration in the body. Myostatin concentration rises with age, adding to increased muscle & bone loss (sarcopenia & osteopenia).
- Physical activity plays an important role in myostatin regulation, where a decrease of physical activity achieved by daily step reduction increased myostatin expression. Myostatin levels were found to decrease acutely after various exercise types (ultramarathon, aerobic, resistance and high-intensity training). Resistance training, especially in older adults is an excellent way to inhibit myostatin.
- Studies demonstrated that circulating myostatin levels are increased in metabolically unhealthy individuals, i.e. obese and suffering from insulin resistance. Myostatin concentration was found to be higher in prediabetes and the highest in type 2 diabetes mellitus.
- Various drugs can influence the expression and level of myostatin in muscles and serum. Studies suggest the potential impact of treatment with various substances (such as acetaminophen, losartan, insulin, denosumab, dapagliflozin or glucocorticoids) on the serum myostatin level.
Considerable improvement in understanding of how myostatin activity and signalling is regulated has led numerous pharmaceutical and biotechnology companies to develop promising myostatin inhibitors including antibodies, soluble receptors, and natural antagonists that have successfully progressed through early stages of clinical testing. Clearly, many of these approaches promote robust increases in muscle and bone mass; however, they differ in efficacy and safety. Myostatin inhibitor drugs have the potential to be greatly beneficial against muscle wasting diseases and disorders, yet to date, have been highly ineffective.
MYOSTATIN AND SPORTS
The ability of myostatin to limit the growth of muscle mass immediately attracted the attention of sports researchers, specially sports that require muscle strength and mass. In fact, the genetic predisposition to gain muscle mass is due to the low expression of myostatin, which is advantageous in the improvement of strength.

According to World Anti-Doping Agency (WADA), gene doping is the non-therapeutic manipulation of cells, genes, genetic elements or modulation of gene expression with the aim of increasing athletic ability. That is the reason myostatin inhibition is banned by WADA.
But how does myostatin work in your body?
When you perform power and strength based exercises, it causes mechanical damage to your muscle fibres. After a certain period of time, the balance shifts toward protein synthesis and, as a result, the volume and strength of skeletal muscles, increases.
These processes release active myostatin, which affects satellite cells and fibroblasts located near the damaged area. Myostatin can cause protein degradation in myofibrils, which are important for the normal functioning of muscle fibres as they remove unnecessary, wasted proteins from the muscle cells.
That’s why, myostatin is one of the main factors associated with muscle atrophy. In fact, studies showed that, prolonged sedentary lifestyle, leads to dramatic increase in the levels of myostatin gene.
Muscle cells (or muscle fibres) contain hundreds to thousands of little nuclei/Myonuclei. The nuclei/myonuclei are little control centres, allowing for the rapid, and coordinated growth and repair of muscle tissue.
Sustained hypertrophy of muscles requires the addition of new nuclei by muscle stem cells (satellite cells). More nuclei means more efficient growth and repair signals, which are required to adequately meet the growing cell’s/muscle fibre’s needs. Thus, with increase in muscle size, the number of nuclei increases.
Also, we need to understand that our muscle cells can’t produce nuclei. Instead they take it from another type of cell called – stem cell. Stem cells are special cells that can be developed into many different types of cells in the body.
There are many different kinds of stem cells in the body, but the kind most involved in muscle growth are referred to as satellite cells. These cells lie dormant near muscle cells and are recruited as needed to help heal and repair damaged muscle fibres.
Once called upon, satellite cells attach themselves to damaged muscle cells and donate their nuclei, which aids in repair and increases the cells’ potential for more size and strength. (This is one of the processes that causes your muscles to get bigger and stronger when you lift weights.)
Primarily, microtrauma in a single muscle fibre acts as a stimulus for the proliferation of satellite cells in adult organisms. When these cells are activated and emerge from a dormant state, genes characteristic of myoblasts are also activated. Therefore, satellite cells become myoblasts that migrate to the damaged areas of muscle tissue and depending on the degree of damage, either merge with the damaged muscle fibre (hypertrophy) or merge with each other, thus creating new fibres (hyperplasia).
Myostatin negatively regulates the activation of resting satellite cells, hindering their development. Such inhibitory effects are necessary for normal muscle regeneration as a premature fusion of satellite cells with myofibrils can impair muscle fibre functions.
Apart from muscles, myostatin also affects ligaments, tendons & bone density. Weightlifting and speed-strength sports athletes with high skeletal muscle strength often have tendon injuries as their muscle strength exceeds their endurance. During strength training, fibroblasts proliferate, collagen synthesis increases and the cross-sectional area of the tendons increases to make them stiffer. This allows the tendons to withstand high intensity physical loads and reduce the risk of damage to them.
Myostatin can change the mechanical properties of tendons by impairing their ability to stretch. The exact mechanisms of the effect of myostatin on tendons and ligaments are still unknown, but it is assumed that myostatin effect the expression of Type I collagen. Similarly, the effect on bone density is still being studied, but it is assumed that myostatin inhibition will also help in increasing bone density, just like muscle mass.
MYOSTATIN SUPPLEMENT REALITY
There have been a number of supplements claiming to inhibit myostatin in humans and skyrocket your muscle growth, but the studies are very scarce when it comes to oral supplementation and myostatin gene inhibition.

Creatine has shown some positive effects in this regard. Studies have shown that resistance training causes significant decrease in serum levels of myostatin, and creatine supplementation in conjunction with resistance training lead to greater decreases in serum myostatin.
Another compound stated to reduce myostatin levels is Epicatechin. Epicatechin is a naturally occurring flavanol found in cacao and green tea and has been orally ingested safely by humans for many centuries. They have strong antioxidant properties.
A study, investigated the effects of resistance training and epicatechin supplementation on muscle strength, and myostatin in 62 older adults with sarcopenia. Participants were divided into resistance training (RT), epicatechin (EP), resistance training + epicatechin (RT+EP), and placebo (PL). The greatest increase in strength and decrease in myostatin was found in the RT+EP group.
However, another study, determined if cycling exercise combined with epicatechin supplementation was more effective at increasing training adaptations than cycling combined with a placebo. Results indicated that epicatechin supplementation does not affect myostatin gene expression or anaerobic training adaptations.
Spirulina has also shown to reduce myostatin levels, but the studies are too mixed and preliminary to comment on.
However, one of the biggest scams currently in trend is the supplements which are falsely marketed to dramatically reduce your myostatin levels, without even a single evidence proving their efficacy. Worse, there are company funded studies showing absolutely absurd and nonsensical figures.
For e.g. a study on Myo-T12 supplement of the brand MHP, showed a 46% reduction in myostatin in just 12-18hrs, with just a single serving. At this rate, you would simply finish your myostatin levels in couple of days, and turn into a muscle monster.



